Published with Blogger-droid v2.0.10
Sunday, February 17, 2013
Wednesday, June 6, 2012
Drying vs tempreature
After getting a bunch of new samples, I decided to delve in the drying methods of the chiku. Previously, I've tried air drying out in the open and cabinet drying, both in room temperature. But there are a few setbacks in this method, such as the long time that increases the susceptibility to airborne fungi contamination. Thus, as I found out in the previous post, there are many temperatures and settings that can be used to dry the cut fruit. Therefore I studied different temperatures that is most effective to dry a sample, as in the research of Chien et al, 2009.
Three samples of similar maturity (around 1kg per temperature) is peeled, sliced and oven-dried into constant weight. Three temperatures are used--50, 60, and 70. The time of drying, yield, and properties is as follows.
According to the Chien study, higher temperatures can give better antioxidant activity. This is the exact opposite of some other fruit drying results such as Larrauri et al, (1997), Garau et al (2007), and Shiau, Wu and Chang (2012); but similar to results of Madrau et al, (2008), where its antioxidant components slightly increase. Although in this study, it claims that 60C does not change much of the fruits' properties.
Therefore, I shall use the middle tempreature of 60C for drying. Although I need to make sure there are less gummy samples after drying.
Tuesday, May 29, 2012
Extraction Types
During the course of the fruit extraction, I've been experimenting with two extraction types lately, one being water extraction and the other being ethanol 95%. When tested with the same five microbes, these are the results:
1) Water extraction
2) Ethanol extraction
From these tests I can conclude that both extract had somewhat of an inhibition activity towards microbes tested. A clear ring is seen around the disks for both extract types (except conc 25% and below). However, water extraction is not very effective in showing anti m/o activity. There is a huge difference in the two activities, even of similar fruits with similar maturity. According to the S/V, since ciku is full of polyphenols and other antioxidants (non-polar solvent soluble), those activities may be more observable in non-polar solvent.
One other reason that proved this is the fact that Ciku has very high antioxidant capacity. L-ascorbic acid equivalent antioxidant capacity, AEAC, is the highest compared to other local fruits such as starfruit, guava, salak, pineapple and so on.
 |
| from Leong and Shui, 2001 |
In this experiment above, the ciku used is unripe. Fruit is peeled and blended, and mixed into 50% ethanol at 1:10 w/v, and centrifuged to obtain supernatant. This is different than the method I encountered which used ripe fruits (H. N. Sin et al, 2006) . However, after many unsuccessful attempts with ethanol extract (very little antimicrobial activity), I may conclude that ethanol may not be fully optimized for these kind of fruits. I may need other types of extracting solvent. A few suggestions are hexane, acetone and methanol.
One research delved on the various types of extraction, and chose acetone as the preferred solvent (Shui, Wong, and Leong, 2004). Acetone 50% is used directly to extract pulp of ciku for 45 minutes at 90C. Then it is rapidly cooled to room temperature before centrifugation at 14000g. This extract is used for spectrophotometry testing of TPC. Another research (Chong et al, 2005) also uses acetone (70%), but after it the ciku was homogenized it in ultrasonic bath for 30 minutes before centrifuging it for 15 min at 5000rpm. This is different than Ma et al, (2003), who uses methanol for ciku extraction and later fractionate it using hexane and ehtyl acetate.
Another possible error (while doing ethanol extraction) is the drying part. Previously I used "room temperature drying", i.e. 26-28C, to dry the ciku before extraction as specified by Jayakumar and Kanthimathi (2011). However, it is recently found out by Chong et al (2009), that the higher the drying temperature the higher the phenolics in the ciku.
In this experiment, ciku slices is dried until constant weight, between 15 minutes to 7 hours. Therefore, higher temperature can be used to dry the ciku; with only the time period is of concern.One research delved on the various types of extraction, and chose acetone as the preferred solvent (Shui, Wong, and Leong, 2004). Acetone 50% is used directly to extract pulp of ciku for 45 minutes at 90C. Then it is rapidly cooled to room temperature before centrifugation at 14000g. This extract is used for spectrophotometry testing of TPC. Another research (Chong et al, 2005) also uses acetone (70%), but after it the ciku was homogenized it in ultrasonic bath for 30 minutes before centrifuging it for 15 min at 5000rpm. This is different than Ma et al, (2003), who uses methanol for ciku extraction and later fractionate it using hexane and ehtyl acetate.
Another possible error (while doing ethanol extraction) is the drying part. Previously I used "room temperature drying", i.e. 26-28C, to dry the ciku before extraction as specified by Jayakumar and Kanthimathi (2011). However, it is recently found out by Chong et al (2009), that the higher the drying temperature the higher the phenolics in the ciku.
Penanda buku:
extraction,
research,
solvent
Thursday, April 19, 2012
2. Antimicrobial activity, minimum inhibitory conc.
The continuation of the research has focused
on the two most significantly inhibited microbes, the E.coli (EC) and the
L.monocytogenes (LM). For the other three, last week’s experiment is repeated
on them, using fresh culture.
2.1 For EC and LM, three concentrations are
prepared; 100%, 50% and 25%. The original sample is diluted until the desired
concentration.
Result is as follows:
a) LM
Conc.
|
50
|
25
|
1
|
12
|
15
|
2
|
12
|
0
|
3
|
13
|
0
|
AVERAGE
|
12.3
|
15
|
b) EC
Conc.
|
50
|
25
|
1
|
0
|
12.5
|
2
|
0
|
12
|
3
|
0
|
12
|
AVERAGE
|
0
|
12.16667
|
Main problem: Sample is too viscous and sticky
to be removed using a pipette. Therefore solvent is poured in and shaken as
much as possible to dissolve the sample. But not all is dissolved, some remain
sticking to the base of the container.
Therefore it is difficult to gauge the real
inhibitory activity for 25% and 50% since there might actually be lower concentration
of ciku compound in diluted sample.
2.2 For Retest of antimicrobial susceptibility
(SA, STY and I), result is as follows:
Take note that if a “ring” is observed for streptococci
types, it is deemed resistant regardless of the clear zone size, because of
the beta-lactamase produced by the m/o.
Suggestion:
to do it in a spread plate method, using cotton swabs, to make sure the m/o spreads properly.
Also, look into other journals, to see what kind of extract is used--eg. methanol and hexane, since the viscosity may be attributed to the solvent.
Suggestion:
to do it in a spread plate method, using cotton swabs, to make sure the m/o spreads properly.
Also, look into other journals, to see what kind of extract is used--eg. methanol and hexane, since the viscosity may be attributed to the solvent.
Penanda buku:
antimicrobial,
mic,
research,
resistant ring
Sunday, April 15, 2012
Antimicrobial test: Result!
In continuation with last week's experiment result (which proved that my current extract is contaminated), another extract is used to repeat the experiment. This time I used an older extract, which is much more dark, concentrated and viscous (therefore cannot be poured using a pipette).
The method is still the same, that is:
Prepare a 24 hour culture suspension of the selected microbes: S. aureus, E. Coli, L. monocytogenes, S. typhi, and S. I.
An agar-well test is made using the selected microbes. Three holes of .7mm are plugged out on each plate and poured with samples. (Samples are used at 100% conc to see their effectiveness first) The plates are incubated 18hr in 37C.
Results are in as follows:
Therefore, after much measurement, we have:
Looking at I plate sets, the zone are not that clear, but the microbe growth is not that vigorous either. There are spots of microbe in the "zone" around the plates. This either due to 1) microbes in I are slow-growing (not so) or 2) it's very effective that no microbe has grown. The little growth seen are probably lucky few contaminant/resistant ones.
In conclusion, the sample is very effective on L.monocytogenes, E.coli, and not effective on S.aureus and S.typhi. More test should be done to confirm I.
The method is still the same, that is:
Prepare a 24 hour culture suspension of the selected microbes: S. aureus, E. Coli, L. monocytogenes, S. typhi, and S. I.
An agar-well test is made using the selected microbes. Three holes of .7mm are plugged out on each plate and poured with samples. (Samples are used at 100% conc to see their effectiveness first) The plates are incubated 18hr in 37C.
Results are in as follows:
 |
| ST-no zone |
 |
| SA-no zone |
 |
| LM- clear zone |
 |
| EC-clear zone |
 |
| I-not much growth, but not very clear zone |
 |
| Average inhibition zone, in mm |
In conclusion, the sample is very effective on L.monocytogenes, E.coli, and not effective on S.aureus and S.typhi. More test should be done to confirm I.
Penanda buku:
antimicrobial,
ciku,
cup well method,
discussion,
research,
success
Tuesday, April 10, 2012
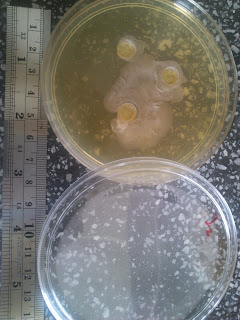
What went wrong?
A good trait of a scientifically-trained person is to investigate a result that goes differently than expected. Now, what is to do, for example, if the result seems neither bad nor good?
The case in hand--this is an antimicrobial test using the Agar-Well method. 24-hour cultured microbes are grown in plates using pour agar method. 100 microliter of sample extract is poured into wells of 0.7mm diameter. Then, it is incubated for 18 hours. The expected result is an inhibition zone, that is a clear zone with no microbe growth around the well of the sample.
The result that came out isn't as expected--an inhibition zone is shown all right; but it is not a clear zone. Rather, it seems very opaque. As follows.
So, after much pondering, possible explanation is as follows:
1) the sample really spread outside the well, creating an inhibition zone.
2) that wasn't the sample, it was a living, growing microbe.
therefore, this hypothesis is put to the test by taking a bit from the "inhibition zone" and streaking it on a new plate:
A growth of similar shape and color occurs, which tells me this is a microbe. So the (2) reason is accepted.
Then, looking deeper, the second reason has two possible outcomes, that is:
a) the sample is contaminated from somewhere else.
b) the sample has it's own microbe that has inhibitory properties.
So, to test this, is to take a swab of the original sample straight from the bottles, for every batch.
The swab is streaked on a nutrient agar and left for 18 hours. If not all the sample shows the growth, (a) is true. But if every batch shows the same microbe, (b) is true.
The result is shown as below:
Well, only one sample plate shown to have the microbe--the 6th March sample used in the agar-well test. Therefore, it is proven that this is a one-off contamination and not a special microbe.
Another thing to add--during the agar well test though, in some other plates, the growth inhibition effect does not show. it can be explained, since probably the microbe in the sample is unable to inhibit the culture's microbes.
So in conclusion, one of the sample was contaminated with a microbe that is selectively inhibiting. May be useful, maybe not. We'll see.
I guess that is how a student's mind should work eh. Later guys :)
The result that came out isn't as expected--an inhibition zone is shown all right; but it is not a clear zone. Rather, it seems very opaque. As follows.
So, after much pondering, possible explanation is as follows:
1) the sample really spread outside the well, creating an inhibition zone.
2) that wasn't the sample, it was a living, growing microbe.
therefore, this hypothesis is put to the test by taking a bit from the "inhibition zone" and streaking it on a new plate:
A growth of similar shape and color occurs, which tells me this is a microbe. So the (2) reason is accepted.
Then, looking deeper, the second reason has two possible outcomes, that is:
a) the sample is contaminated from somewhere else.
b) the sample has it's own microbe that has inhibitory properties.
So, to test this, is to take a swab of the original sample straight from the bottles, for every batch.
The swab is streaked on a nutrient agar and left for 18 hours. If not all the sample shows the growth, (a) is true. But if every batch shows the same microbe, (b) is true.
The result is shown as below:
Well, only one sample plate shown to have the microbe--the 6th March sample used in the agar-well test. Therefore, it is proven that this is a one-off contamination and not a special microbe.
Another thing to add--during the agar well test though, in some other plates, the growth inhibition effect does not show. it can be explained, since probably the microbe in the sample is unable to inhibit the culture's microbes.
So in conclusion, one of the sample was contaminated with a microbe that is selectively inhibiting. May be useful, maybe not. We'll see.
I guess that is how a student's mind should work eh. Later guys :)
Penanda buku:
agar well,
antimicrobial,
ciku,
error,
investigation,
research,
what went wrong
Tuesday, March 20, 2012
Apakah itu "Factoring agent"?
So tadi semasa sedang urus-mengurus PO dan RO (tersilap sebut PO dekat staff, pening dia cari borang, rupa-rupanya PO. haha!) adalah terkeluar persoalan mengenai Factoring Agent.
Awalnya saya sangkakan bahawa factoring agent ini sejenis bentuk formula matematik (factoring = pemfaktoran). Rupa-rupanya ia adalah istilah perniagaan untuk ejen orang tengah dalam urusniaga.
Bayangkan suatu situasi: Anda menjadi pembekal kepada institusi kerajaan. Dan seperti yang anda tahu, institusi kerajaan ni banyak pula karenah birokrasinya (dalam bahasa inggeris dipanggil red tape). Duit nak bayar memang ada tapi, bayarannya lambat kredit. Jika anda sebuah syarikat kecil yang baru nak naik, dan pusingan modal anda lambat, ini boleh menyebabkan duit anda "terikat" tanpa aliran tunai. Tentu susah bukan?
Jadi salah satu cara mengatasinya ialah mendapatkan sebuah ejen, Factoring Agent. Mereka akan menjadi orang tengah antara anda dan pelanggan, di mana pelanggan akan membayar kepada mereka, dan mereka akan membayar kepada anda--dengan sedikit upah, tentunya. Contohnya begini.
Pihak Jabatan A memesan barang. Anda pun menghantar barang.
Anda pun mengeluarkan invois untuk Jabatan A itu. Tapi, invois itu dibayar kepada akaun Ejen B. Sebaik saja invois diberikan. Ejen B membayar sebahagian daripada nilai invois itu kepada anda, WALAUPUN Jabatan A tak bayar lagi duit (kira sekarang Jabatan A berhutang kepada Ejen B).
Apabila Jabatan A bayar kepada Ejen B mengikut invois tersebut, Ejen B akan memberi baki wang selebihnya kepada anda. Tapi dia tolak sikit untuk untung. Contoh kalau harganya 100 ringgit, dia ambil 5 ringgit buat simpanan sendiri (diorang pun nak cari makan juga kan. haha!).
Apa akan jadi kalau customer cabut lari tak bayar? Ha, itu kena tengok pula perjanjian dengan Ejen B. Biasanya anda yang kena tanggung (recourse agreement), tapi ada juga yang mereka akan tanggung (non-recourse agreement). Kalau mereka yang tanggung tentunya untung yang diambil nanti agak banyak, tetapi anda juga selamat daripada pelanggan yang melarikan diri.
Begitulah adanya Factoring Agent. Kelebihannya ialah memudahkan dokumentasi akaun kerana kita berurusan dengan sorang ejen sahaja. Satu lagi adalah ia membebaskan aliran tunai dan membolehkan kita membeli lebih banyak inventori dan menjalankan projek jangka pendek. Jadi bagi syarikat yang mengutamakan pertumbuhan memang sesuai untuk membuat Factoring.
Apa-apapun saya tidaklah mahir sangat, ini barang dari google sahaja. Dapatkan maklumat lebih lanjut daripada perancang kewangan profesional. Assalamualaikum!
Penanda buku:
definisi,
ejen pengilangan,
factoring agent,
finance,
perkongsian,
syarikat kecil
Subscribe to:
Posts (Atom)